

Investor Presentation January 11, 2021 Nasdaq: ATRA Exhibit 99.1

Forward-Looking Statements This presentation and the accompanying oral presentation contain forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical facts contained in this presentation, including statements regarding our future results of operations and financial position, business strategy, product candidates, regulatory approvals, the initiation, timing, progress and results of preclinical studies and clinical trials and our research and development programs, ability to sell, manufacture or otherwise commercialize our product candidates, research and development costs, timing and likelihood of success, plans and objectives of management for future operations, any royalty payments, and our ability to obtain and maintain intellectual property protection for our product candidates, are forward-looking statements. These statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. These forward-looking statements are subject to risks and uncertainties, including those discussed in Atara Biotherapeutics' filings with the Securities and Exchange Commission (SEC), including in the “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” sections of the Company’s most recently filed periodic reports on Form 10-K and Form 10-Q and subsequent filings and in the documents incorporated by reference therein. Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified and some of which are beyond our control, you should not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. Certain information contained in this presentation and statements made orally during this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and Atara's own internal estimates and research. While Atara believes these third-party studies, publications, surveys and other data to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, no independent source has evaluated the reasonableness or accuracy of Atara’s internal estimates or research and no reliance should be made on any information or statements made in this presentation relating to or based on such internal estimates and research. The content of this presentation is subject to copyright, which will be asserted by Atara and no part of this presentation may be reproduced, stored in a retrieval system, or transmitted in any form or by any means without prior permission in writing from Atara.

Ola PTLD champion Ayden PTLD champion Jon PTLD champion Doug PTLD champion Jessica PTLD champion 1982-2019 Atara mourns the loss of Jessica, who passed away on September 25, 2019 while awaiting a new heart and kidney transplant. Her memory continues to fuel our urgency in developing new therapies for devastating diseases. Our mission is to transform the lives of patients with serious diseases through pioneering science, teamwork and a commitment to excellence Pioneering Off-the-Shelf, Allogeneic T-cell Immunotherapies Dan MS champion

Highly Experienced Executive Team Dedicated to Transforming the Lives of Patients Kristin Yarema, Ph.D. Chief Commercial Officer Pascal Touchon President and Chief Executive Officer Joe Newell Chief Operations Officer AJ Joshi, M.D. Chief Medical Officer Utpal Koppikar Chief Financial Officer Jakob Dupont, M.D. Head of Research and Development

We Are a Leading Allogeneic T-Cell Immunotherapy Company ATA188: Potentially Transformative MS Treatment in Randomized Controlled Trial Tab-cel®: First-In-Kind, Late-Stage, Oncology Program Differentiated Allogeneic Cell Therapy Platform Next-Gen Allogeneic CAR T Portfolio, Validated by Bayer Collaboration on Mesothelin-Targeted CAR T Scalable EBV T Cell platform and technologies to develop multiple allogeneic cell therapies BLA completion expected in Q3 2021, underpinned by strong clinical data Competitive programs designed to address current limitations of autologous and allogeneic CAR T Working towards placebo-controlled data, expected within ~2 years, to enable pivotal studies and partnering opportunities Advanced process science and wholly owned pre-commercial manufacturing capabilities attractive to potential partners Proven Technical Capabilities EBV = Epstein-Barr Virus; BLA = Biologics License Application; BTD = Break Through Designation
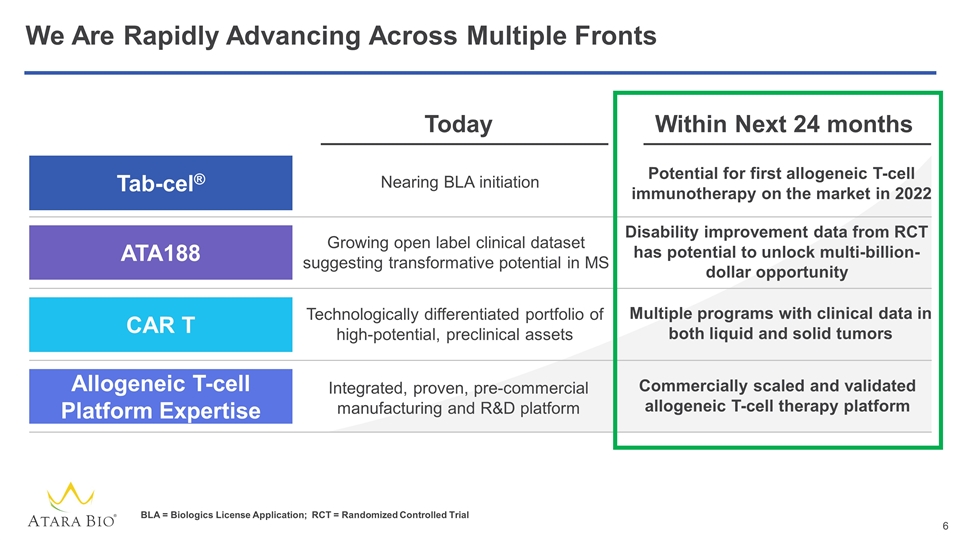
We Are Rapidly Advancing Across Multiple Fronts Today Within Next 24 months Tab-cel® ATA188 CAR T Allogeneic T-cell Platform Expertise Nearing BLA initiation Potential for first allogeneic T-cell immunotherapy on the market in 2022 Growing open label clinical dataset suggesting transformative potential in MS Disability improvement data from RCT has potential to unlock multi-billion-dollar opportunity Technologically differentiated portfolio of high-potential, preclinical assets Multiple programs with clinical data in both liquid and solid tumors Commercially scaled and validated allogeneic T-cell therapy platform Integrated, proven, pre-commercial manufacturing and R&D platform BLA = Biologics License Application; RCT = Randomized Controlled Trial
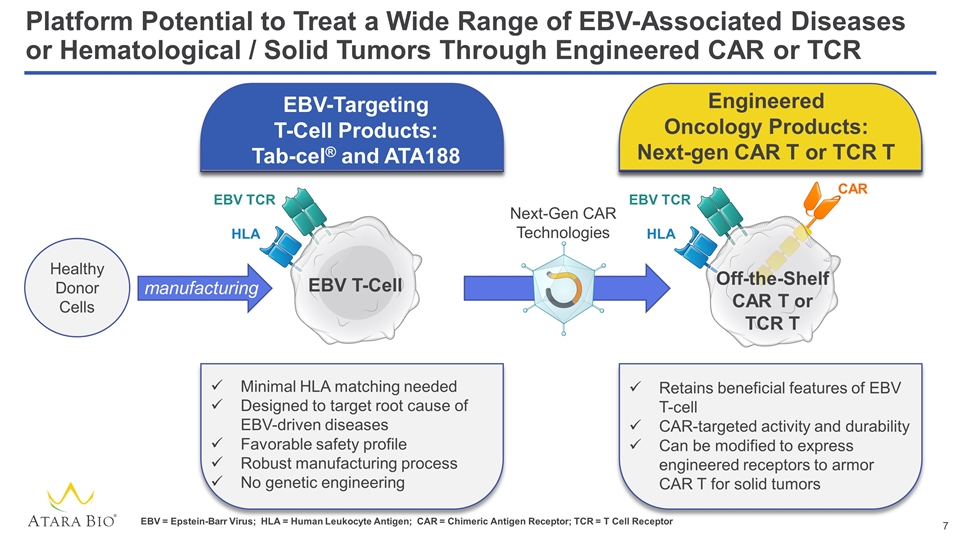
Platform Potential to Treat a Wide Range of EBV-Associated Diseases or Hematological / Solid Tumors Through Engineered CAR or TCR Healthy Donor Cells Minimal HLA matching needed Designed to target root cause of EBV-driven diseases Favorable safety profile Robust manufacturing process No genetic engineering EBV T-Cell Off-the-Shelf CAR T or TCR T Next-Gen CAR Technologies Retains beneficial features of EBV T-cell CAR-targeted activity and durability Can be modified to express engineered receptors to armor CAR T for solid tumors EBV-Targeting T-Cell Products: Tab-cel® and ATA188 Engineered Oncology Products: Next-gen CAR T or TCR T EBV TCR HLA EBV TCR HLA CAR manufacturing EBV = Epstein-Barr Virus; HLA = Human Leukocyte Antigen; CAR = Chimeric Antigen Receptor; TCR = T Cell Receptor
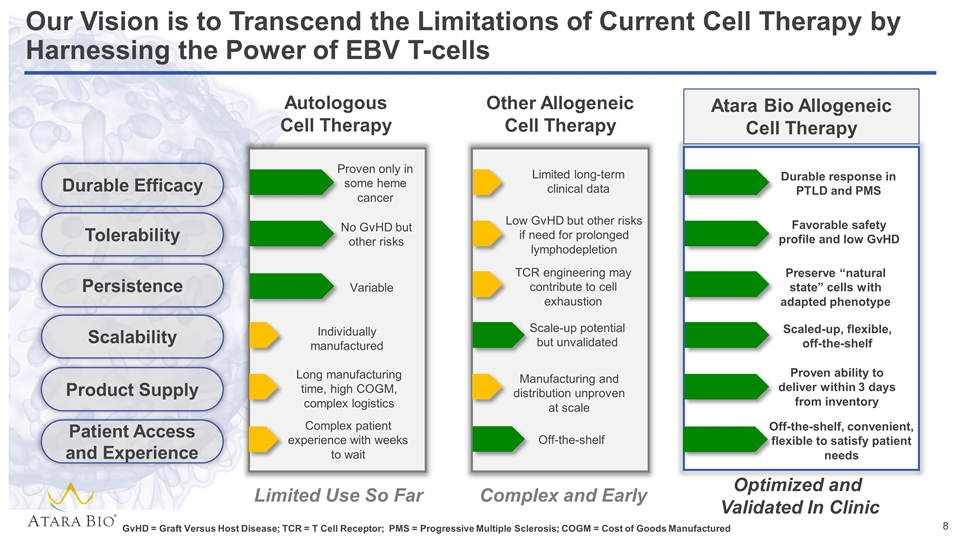
Our Vision is to Transcend the Limitations of Current Cell Therapy by Harnessing the Power of EBV T-cells Autologous Cell Therapy Other Allogeneic Cell Therapy No GvHD but other risks Individually manufactured Long manufacturing time, high COGM, complex logistics Low GvHD but other risks if need for prolonged lymphodepletion Durable response in PTLD and PMS Favorable safety profile and low GvHD Scale-up potential but unvalidated Variable TCR engineering may contribute to cell exhaustion Preserve “natural state” cells with adapted phenotype Scaled-up, flexible, off-the-shelf Manufacturing and distribution unproven at scale Proven ability to deliver within 3 days from inventory Limited long-term clinical data Proven only in some heme cancer Limited Use So Far Complex and Early Optimized and Validated In Clinic Durable Efficacy Tolerability Persistence Scalability Product Supply Patient Access and Experience GvHD = Graft Versus Host Disease; TCR = T Cell Receptor; PMS = Progressive Multiple Sclerosis; COGM = Cost of Goods Manufactured Complex patient experience with weeks to wait Off-the-shelf Off-the-shelf, convenient, flexible to satisfy patient needs Atara Bio Allogeneic Cell Therapy
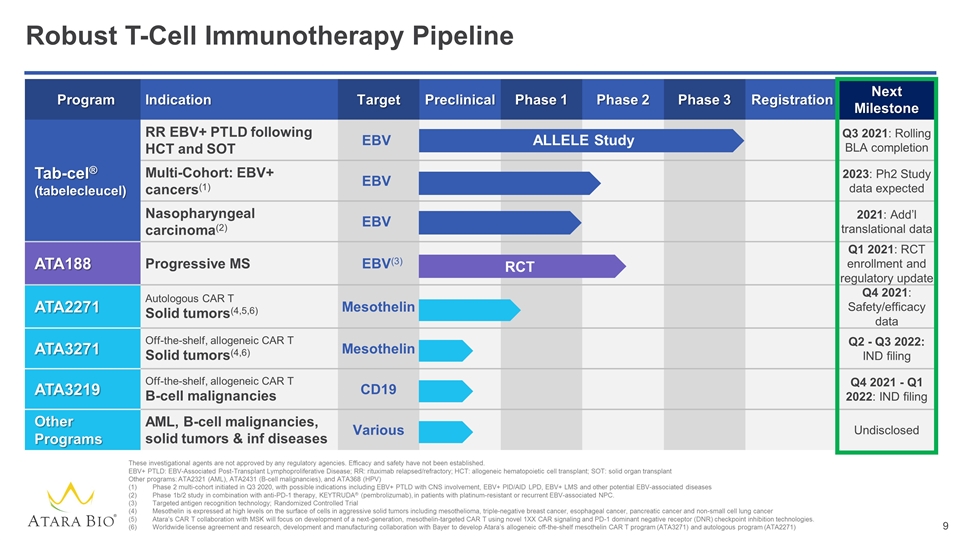
Program Indication Target Preclinical Phase 1 Phase 2 Phase 3 Registration Next Milestone Tab-cel® (tabelecleucel) RR EBV+ PTLD following HCT and SOT EBV Q3 2021: Rolling BLA completion Multi-Cohort: EBV+ cancers(1) EBV 2023: Ph2 Study data expected Nasopharyngeal carcinoma(2) EBV 2021: Add’l translational data ATA188 Progressive MS EBV(3) Q1 2021: RCT enrollment and regulatory update ATA2271 Autologous CAR T Solid tumors(4,5,6) Mesothelin Q4 2021: Safety/efficacy data ATA3271 Off-the-shelf, allogeneic CAR T Solid tumors(4,6) Mesothelin Q2 - Q3 2022: IND filing ATA3219 Off-the-shelf, allogeneic CAR T B-cell malignancies CD19 Q4 2021 - Q1 2022: IND filing Other Programs AML, B-cell malignancies, solid tumors & inf diseases Various Undisclosed Robust T-Cell Immunotherapy Pipeline These investigational agents are not approved by any regulatory agencies. Efficacy and safety have not been established. EBV+ PTLD: EBV-Associated Post-Transplant Lymphoproliferative Disease; RR: rituximab relapsed/refractory; HCT: allogeneic hematopoietic cell transplant; SOT: solid organ transplant Other programs: ATA2321 (AML), ATA2431 (B-cell malignancies), and ATA368 (HPV) Phase 2 multi-cohort initiated in Q3 2020, with possible indications including EBV+ PTLD with CNS involvement, EBV+ PID/AID LPD, EBV+ LMS and other potential EBV-associated diseases Phase 1b/2 study in combination with anti-PD-1 therapy, KEYTRUDA® (pembrolizumab), in patients with platinum-resistant or recurrent EBV-associated NPC. Targeted antigen recognition technology; Randomized Controlled Trial Mesothelin is expressed at high levels on the surface of cells in aggressive solid tumors including mesothelioma, triple-negative breast cancer, esophageal cancer, pancreatic cancer and non-small cell lung cancer Atara’s CAR T collaboration with MSK will focus on development of a next-generation, mesothelin-targeted CAR T using novel 1XX CAR signaling and PD-1 dominant negative receptor (DNR) checkpoint inhibition technologies. Worldwide license agreement and research, development and manufacturing collaboration with Bayer to develop Atara’s allogeneic off-the-shelf mesothelin CAR T program (ATA3271) and autologous program (ATA2271) ALLELE Study RCT

Dedicated, expandable manufacturing facility Flexibility to produce multiple T-cell and CAR T immunotherapies Designed to meet global regulatory standards Commercial manufacturing validation activities near completion Robust manufacturing process with data confirming potential scale up into perfusion bioreactors enabling biologics-like Cost of Goods Manufactured to supply thousands of patients Product being delivered rapidly to patients across three continents from finished product inventory Our Platform is Nearing Commercial Scale Readiness
Three Strategic Priorities Driving Long-Term Value Tab-cel® ATA188 Next-Gen CAR T BTD program for high unmet need in ultra rare population, with meaningful label expansion potential Compelling efficacy profile in Phase 2 and Phase 3 IA with favorable safety profile Next Step: Initiate rolling BLA and complete BLA submission in Q3 2021 First-in-Kind Allogeneic T-Cell Therapy Preparing for Historic Regulatory Filing High unmet medical need for the ~1 million progressive MS patients worldwide Clinical data support potential to halt or reverse disease progression in progressive MS Next Step: Phase 1a OLE data (ongoing), RCT enrollment and regulatory updates (Q1 2021) Transformative MS Treatment in Randomized Controlled Trial (RCT) Urgent need for new treatment options across solid and liquid tumor indications Portfolio of next-generation CAR T with robust pre-clinical evidence supporting advanced capabilities in solid tumors Next Step: First allogeneic CAR T program IND (Q4 2021 / Q1 2022) Next-Generation Allogeneic CAR T Programs Leveraging EBV T-Cells IA = Interim AnalysisOLE = Open-label ExtensionBLA = Biologics License Application MS = Multiple SclerosisBTD = Break Through DesignationIND = Investigational New Drug CAR = Chimeric Antigen ReceptorEBV = Epstein-Barr Virus

ü ü ü ü Tab-cel® (tabelecleucel) ATA188 Update on long-term clinical data from Phase 1a OLE study in an appropriate forum H1 and H2 2021 Present top-line Phase 1 data for mesothelin-targeted autologous CAR T for patients with advanced mesothelioma Q4 2021 ATA2271 Q3 2021 Complete FDA Biologics License Application (BLA) rolling submission for patients with EBV+ PTLD Q4 2021 Present Phase 3 ALLELE data at an appropriate congress Conduct interim analysis to assess efficacy and safety from Phase 2 randomized, double-blind, placebo-controlled study in patients with progressive forms of MS H1 2022 Submit next-generation off-the-shelf, allogeneic CD-19 targeted CAR T IND for patients with B-cell malignancies Q4 2021 / Q1 2022 ATA3219 Upcoming Key Catalysts Over the Next 24 Months Q4 2021 Submit EU Marketing Authorization Application (MAA) for patients with EBV+ PTLD Complete enrollment of Phase 2 randomized, double-blind, placebo-controlled study in patients with progressive forms of MS H1 2022 Submit next-generation off-the-shelf, mesothelin-targeted allogeneic CAR T IND for patients with advanced mesothelioma Q2 2022 / Q3 2022 ATA3271 H1 2022 Anticipated U.S. approval of BLA for patients with EBV+ PTLD H2 2022 Anticipated EU approval of MAA for patients with EBV+ PTLD
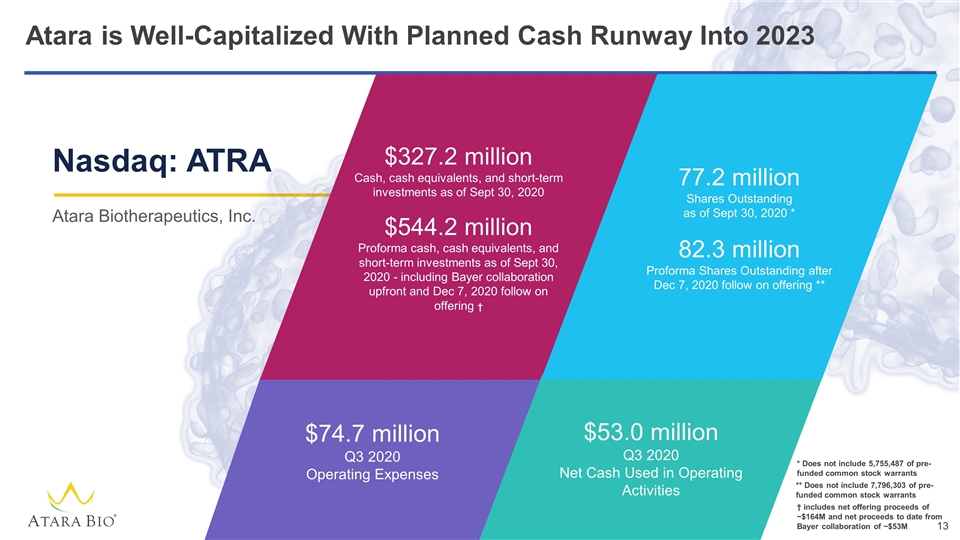
Atara is Well-Capitalized With Planned Cash Runway Into 2023 $327.2 million Cash, cash equivalents, and short-term investments as of Sept 30, 2020 $544.2 million Proforma cash, cash equivalents, and short-term investments as of Sept 30, 2020 - including Bayer collaboration upfront and Dec 7, 2020 follow on offering † $74.7 million Q3 2020 Operating Expenses $53.0 million Q3 2020 Net Cash Used in Operating Activities 77.2 million Shares Outstanding as of Sept 30, 2020 * 82.3 million Proforma Shares Outstanding after Dec 7, 2020 follow on offering ** Nasdaq: ATRA Atara Biotherapeutics, Inc. ** Does not include 7,796,303 of pre-funded common stock warrants * Does not include 5,755,487 of pre-funded common stock warrants † includes net offering proceeds of ~$164M and net proceeds to date from Bayer collaboration of ~$53M

Atara Strategic Priorities to Create Value: Tab-cel® ATA188 CAR T Tab-cel® (tabelecleucel) Investigational T-cell immunotherapy for EBV-associated ultra-rare diseases FDA breakthrough designation and EMA PRIME for EBV+ PTLD

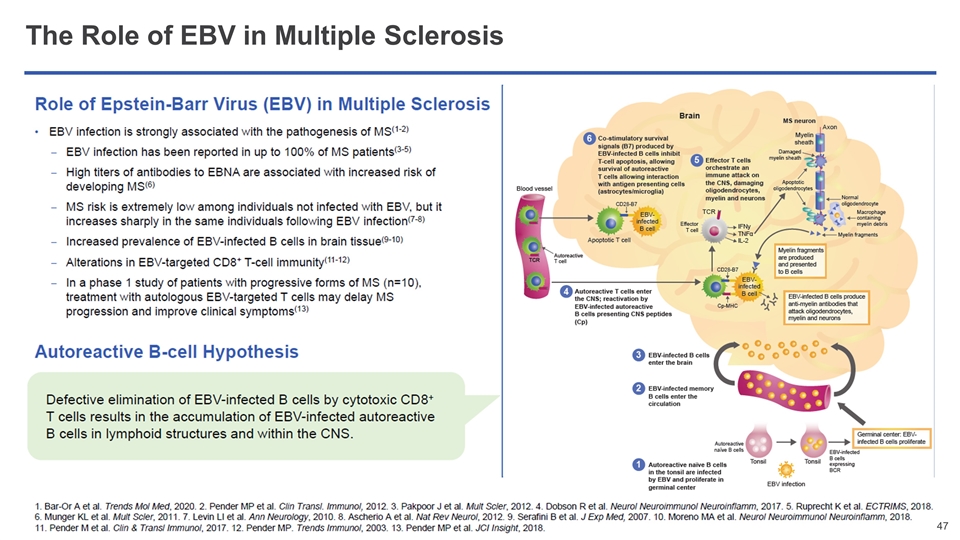
A Common Virus—EBV—Causes Rare and Serious Cancers In Patients With Impaired Immune Function EBV is a common driver of IA-LPDs EBV is a ubiquitous yet typically dormant virus Once infected, healthy patients harbor lifelong infection that is usually kept in check by their immune systems In patients with impaired immune function, uncontrolled growth of EBV-infected cells can lead to lymphomas (IA-LPDs) Such EBV-driven cancers have no approved therapies and poor prognosis with limited life expectancy for patients Patients with impaired immune function include those who have: Conditions requiring immunosuppressive medication (e.g. post-transplant patients, patients with serious autoimmune diseases) Diseases that lower immunity (e.g. HIV) Inborn genetic immune deficiency (e.g. PIDs) EBV: Epstein Barr virus; IA-LPDs: Immunodeficiency-associated lymphoproliferative disorders; PIDs: Primary immunodeficiencies AID = acquired immunodeficiency PID = primary immunodeficiency Phase 2 multicohort study populations include: AID-LPD; PID-LPD; 1L treatment inappropriate PTLD; CNS PTLD; sarcoma, including LMS; viremia with HLH Ph 3 ALLELE study in previously treated EBV+ PTLD Phase 2 multicohort study underway covering six additional patient populations, with aim to expand tab-cel’s label Tab-cel® specifically targets and kills EBV-infected cells, addressing disease at the source Tab-cel has the potential to transform the lives of thousands of patients each year
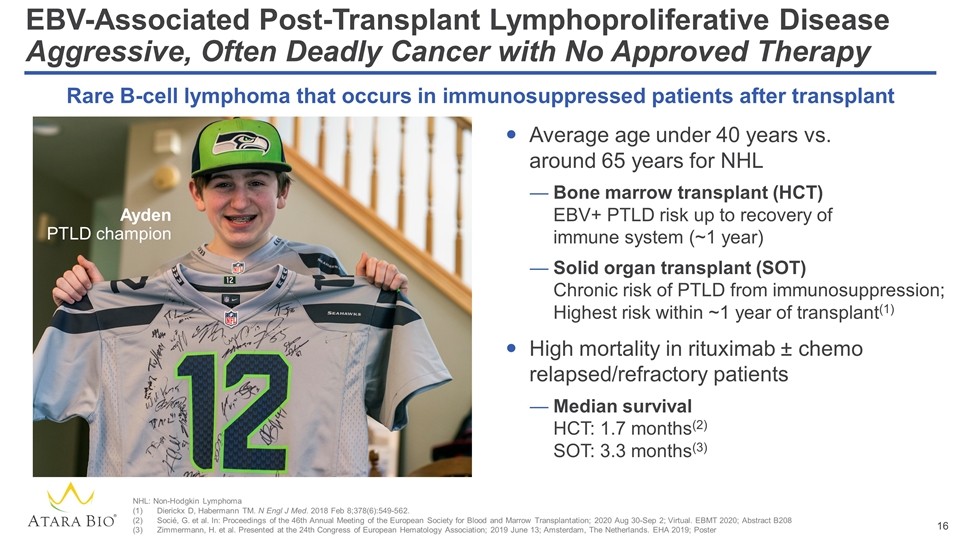
EBV-Associated Post-Transplant Lymphoproliferative Disease Aggressive, Often Deadly Cancer with No Approved Therapy Average age under 40 years vs. around 65 years for NHL Bone marrow transplant (HCT) EBV+ PTLD risk up to recovery of immune system (~1 year) Solid organ transplant (SOT) Chronic risk of PTLD from immunosuppression; Highest risk within ~1 year of transplant(1) High mortality in rituximab ± chemo relapsed/refractory patients Median survival HCT: 1.7 months(2) SOT: 3.3 months(3) Rare B-cell lymphoma that occurs in immunosuppressed patients after transplant NHL: Non-Hodgkin Lymphoma Dierickx D, Habermann TM. N Engl J Med. 2018 Feb 8;378(6):549-562. Socié, G. et al. In: Proceedings of the 46th Annual Meeting of the European Society for Blood and Marrow Transplantation; 2020 Aug 30-Sep 2; Virtual. EBMT 2020; Abstract B208 Zimmermann, H. et al. Presented at the 24th Congress of European Hematology Association; 2019 June 13; Amsterdam, The Netherlands. EHA 2019; Poster Ayden PTLD champion
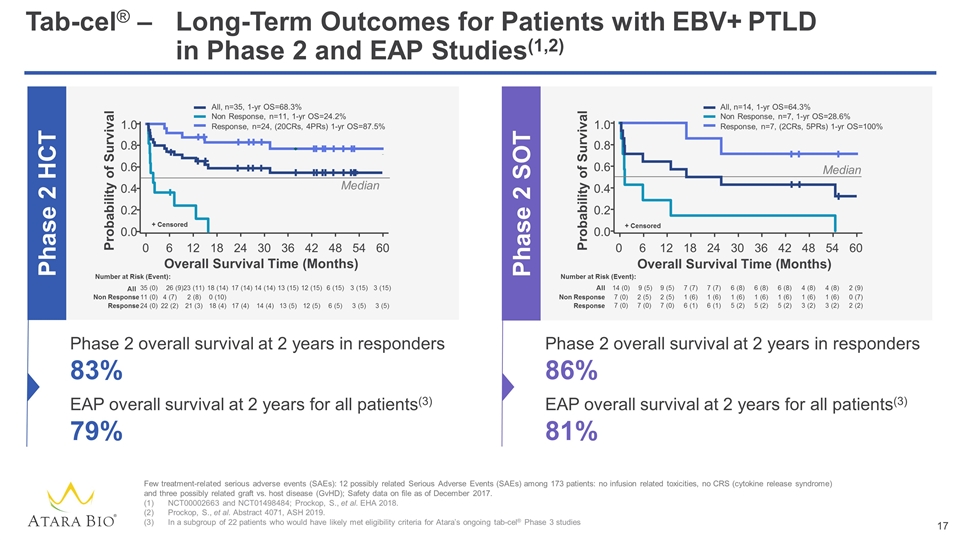
Tab-cel® –Long-Term Outcomes for Patients with EBV+ PTLD in Phase 2 and EAP Studies(1,2) Few treatment-related serious adverse events (SAEs): 12 possibly related Serious Adverse Events (SAEs) among 173 patients: no infusion related toxicities, no CRS (cytokine release syndrome) and three possibly related graft vs. host disease (GvHD); Safety data on file as of December 2017. NCT00002663 and NCT01498484; Prockop, S., et al. EHA 2018. Prockop, S., et al. Abstract 4071, ASH 2019. In a subgroup of 22 patients who would have likely met eligibility criteria for Atara’s ongoing tab-cel® Phase 3 studies Phase 2 SOT Phase 2 HCT All, n=14, 1-yr OS=64.3% Non Response, n=7, 1-yr OS=28.6% Response, n=7, (2CRs, 5PRs) 1-yr OS=100% 14 (0) 7 (7) 9 (5) 9 (5) 7 (7) 6 (8) 6 (8) 6 (8) 4 (8) 4 (8) 2 (9) 7 (0) 1 (6) 2 (5) 2 (5) 1 (6) 1 (6) 1 (6) 1 (6) 1 (6) 1 (6) 0 (7) 7 (0) 6 (1) 7 (0) 7 (0) 6 (1) 5 (2) 5 (2) 5 (2) 3 (2) 3 (2) 2 (2) Overall Survival Time (Months) Median + Censored Number at Risk (Event): All Non Response Response 0 6 12 18 24 30 36 42 48 54 60 0.0 0.6 0.8 0.4 0.2 1.0 Probability of Survival Probability of Survival 0 6 12 18 24 30 36 42 48 54 60 Number at Risk (Event): 35 (0) 18 (14) 23 (11) 26 (9) 17 (14) 14 (14) 13 (15) 12 (15) 6 (15) 3 (15) 3 (15) All 11 (0) 0 (10) 2 (8) 4 (7) Non Response 24 (0) 18 (4) 21 (3) 22 (2) 17 (4) 14 (4) 13 (5) 12 (5) 6 (5) 3 (5) 3 (5) Response + Censored Median All, n=35, 1-yr OS=68.3% Non Response, n=11, 1-yr OS=24.2% Response, n=24, (20CRs, 4PRs) 1-yr OS=87.5% Overall Survival Time (Months) 0.0 0.6 0.8 0.4 0.2 1.0 Phase 2 overall survival at 2 years in responders 83% EAP overall survival at 2 years for all patients(3) 79% Phase 2 overall survival at 2 years in responders 86% EAP overall survival at 2 years for all patients(3) 81%
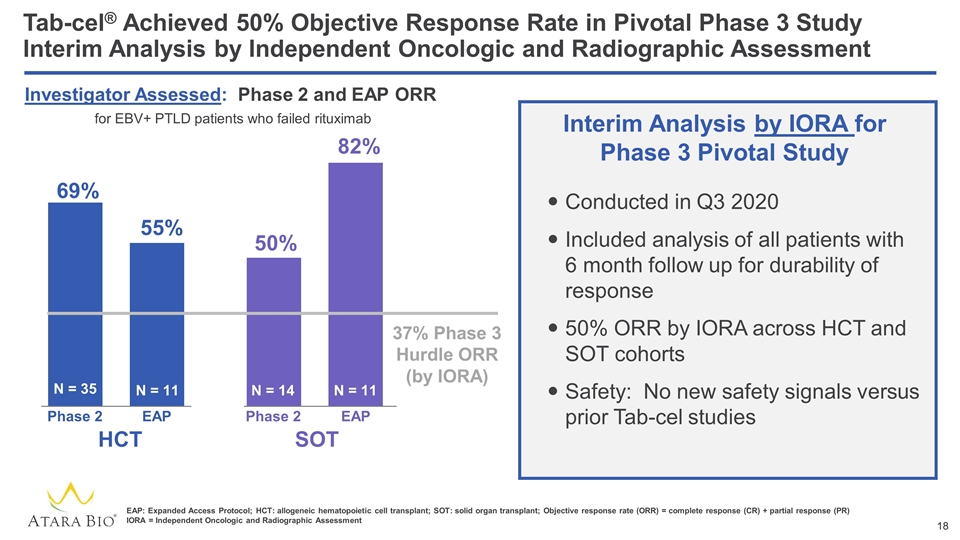
Tab-cel® Achieved 50% Objective Response Rate in Pivotal Phase 3 Study Interim Analysis by Independent Oncologic and Radiographic Assessment 50% N = 35 N = 14 HCT SOT 69% 82% 55% N = 11 N = 11 EAP EAP Phase 2 Phase 2 Investigator Assessed: Phase 2 and EAP ORR for EBV+ PTLD patients who failed rituximab EAP: Expanded Access Protocol; HCT: allogeneic hematopoietic cell transplant; SOT: solid organ transplant; Objective response rate (ORR) = complete response (CR) + partial response (PR) IORA = Independent Oncologic and Radiographic Assessment Conducted in Q3 2020 Included analysis of all patients with 6 month follow up for durability of response 50% ORR by IORA across HCT and SOT cohorts Safety: No new safety signals versus prior Tab-cel studies Interim Analysis by IORA for Phase 3 Pivotal Study 37% Phase 3 Hurdle ORR (by IORA)

Significant Recent Regulatory Progress for Tab-cel® Agreement for rolling BLA submission Agreement on the follow-up period for duration of response needed for currently enrolled patients in pivotal study (ALLELE) Agreement to use prior studies as supportive data in BLA filing Progress on CMC package for BLA Anticipate BLA submission to be completed in Q3 2021 Favorable discussion with PRIME in Q3 2020 on regulatory strategy Pediatric Investigation Plan (PIP) approved in December 2020 On track for MAA submission in H2 2021 BLA = Biologics License Application; MAA = Marketing Authorization Application; CMC = Chemistry, Manufacturing and Control
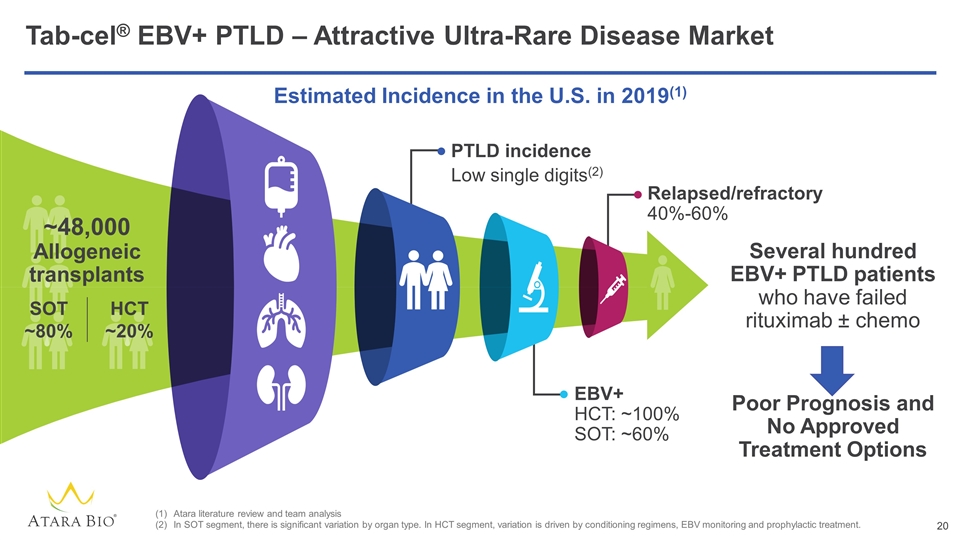
Tab-cel® EBV+ PTLD – Attractive Ultra-Rare Disease Market Atara literature review and team analysis In SOT segment, there is significant variation by organ type. In HCT segment, variation is driven by conditioning regimens, EBV monitoring and prophylactic treatment. Several hundred EBV+ PTLD patients who have failed rituximab ± chemo PTLD incidence Low single digits(2) EBV+ HCT: ~100% SOT: ~60% Relapsed/refractory 40%-60% Estimated Incidence in the U.S. in 2019(1) ~48,000 Allogeneic transplants SOT ~80% HCT ~20% Poor Prognosis and No Approved Treatment Options

Market Dynamics in EBV+ PTLD Are Favorable For Rapid Uptake Of A Transformative Targeted Therapy ~50% of EBV+ PTLD patients fail initial treatment ~2-3 months median survival after failing rituximab with or without chemo Many patients suffer chemo-related side effects, including mortality High rates of diagnosis and treatment for PTLD Guidelines and publications cite need for additional effective options in R/R disease and already include EBV-CTL No approved therapies and today’s options do not specifically target EBV Phase 3 tab-cel® well ahead of a few other therapies being developed in PTLD Ultra-rare, life-threatening and acute disease Significant cost burden to manage PTLD Increasing payor experience covering cell and gene therapies Strong value proposition HIGH UNMET MEDICAL NEED CLEAR PATIENT POPULATION AND CUSTOMER INTEREST PATH TO PATIENT ACCESS STRONG COMPETITIVE POSITION Previously treated EBV+ PTLD is our first planned indication for tab-cel among IA-LPDs Ultra-rare B-cell lymphoma occurring in immunosuppressed post-transplant patients Average age of onset <40 years Several hundred addressable patients per year in US with no approved therapeutic solutions IA-LPDs: Immunodeficiency-associated lymphoproliferative disorders
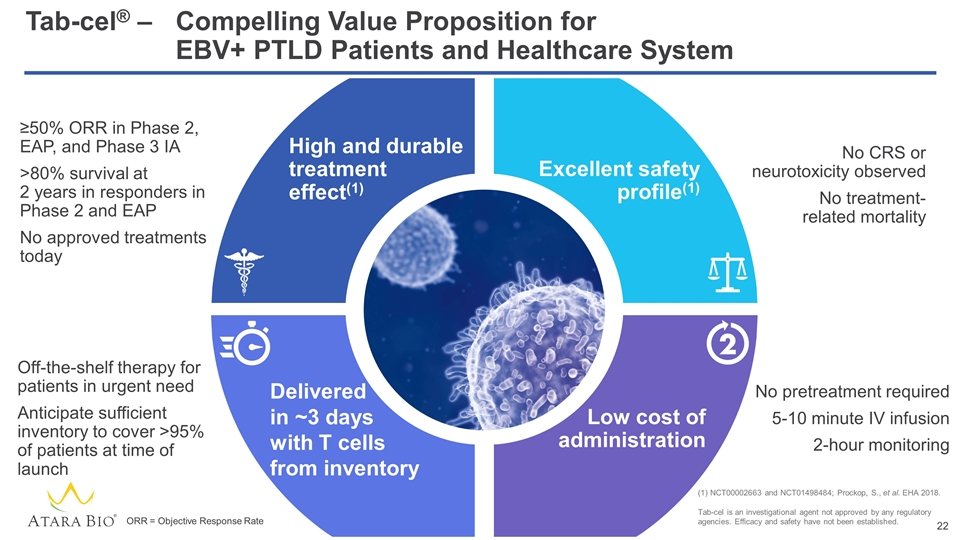
High and durable treatment effect(1) Excellent safety profile(1) Low cost of administration Tab-cel® –Compelling Value Proposition for EBV+ PTLD Patients and Healthcare System No CRS or neurotoxicity observed No treatment- related mortality No pretreatment required 5-10 minute IV infusion 2-hour monitoring Off-the-shelf therapy for patients in urgent need Anticipate sufficient inventory to cover >95% of patients at time of launch ≥50% ORR in Phase 2, EAP, and Phase 3 IA >80% survival at 2 years in responders in Phase 2 and EAP No approved treatments today Delivered in ~3 days with T cells from inventory NCT00002663 and NCT01498484; Prockop, S., et al. EHA 2018. Tab-cel is an investigational agent not approved by any regulatory agencies. Efficacy and safety have not been established. ORR = Objective Response Rate
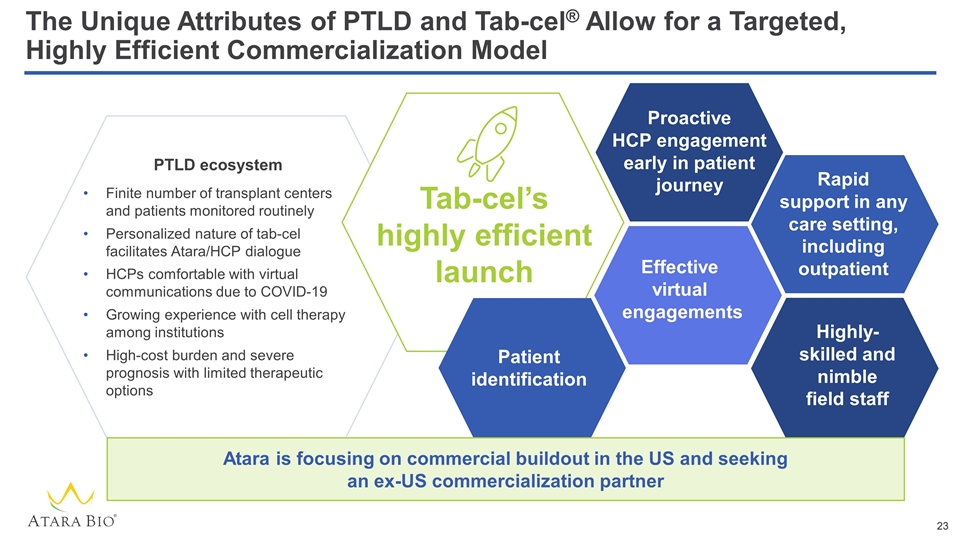
The Unique Attributes of PTLD and Tab-cel® Allow for a Targeted, Highly Efficient Commercialization Model Finite number of transplant centers and patients monitored routinely Personalized nature of tab-cel facilitates Atara/HCP dialogue HCPs comfortable with virtual communications due to COVID-19 Growing experience with cell therapy among institutions High-cost burden and severe prognosis with limited therapeutic options PTLD ecosystem Tab-cel’s highly efficient launch Proactive HCP engagement early in patient journey Patient identification Effective virtual engagements Rapid support in any care setting, including outpatient Highly-skilled and nimble field staff Atara is focusing on commercial buildout in the US and seeking an ex-US commercialization partner
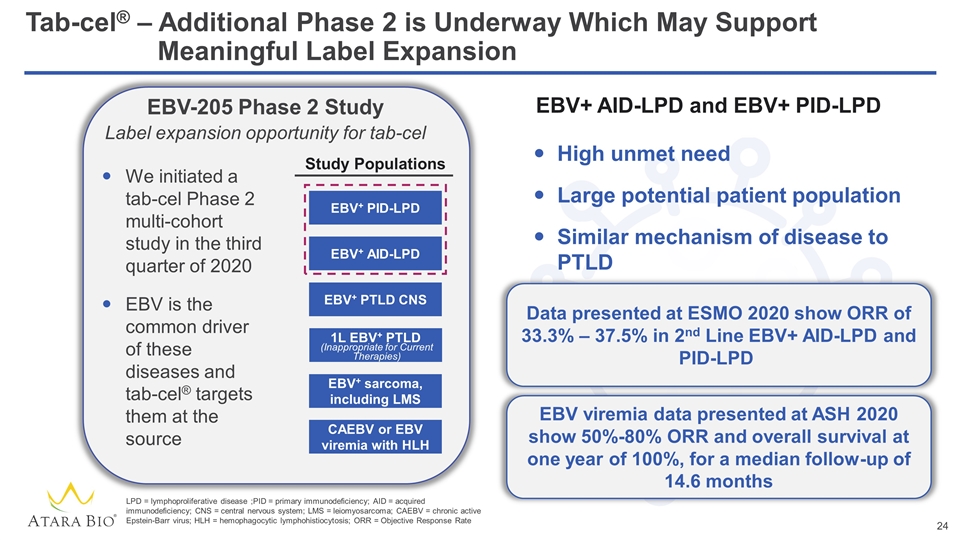
Tab-cel® – Additional Phase 2 is Underway Which May Support Meaningful Label Expansion EBV-205 Phase 2 Study Label expansion opportunity for tab-cel We initiated a tab-cel Phase 2 multi-cohort study in the third quarter of 2020 EBV is the common driver of these diseases and tab-cel® targets them at the source EBV+ PID-LPD EBV+ AID-LPD EBV+ PTLD CNS 1L EBV+ PTLD EBV+ sarcoma, including LMS CAEBV or EBV viremia with HLH (Inappropriate for Current Therapies) LPD = lymphoproliferative disease ;PID = primary immunodeficiency; AID = acquired immunodeficiency; CNS = central nervous system; LMS = leiomyosarcoma; CAEBV = chronic active Epstein-Barr virus; HLH = hemophagocytic lymphohistiocytosis; ORR = Objective Response Rate Study Populations EBV+ AID-LPD and EBV+ PID-LPD High unmet need Large potential patient population Similar mechanism of disease to PTLD Data presented at ESMO 2020 show ORR of 33.3% – 37.5% in 2nd Line EBV+ AID-LPD and PID-LPD EBV viremia data presented at ASH 2020 show 50%-80% ORR and overall survival at one year of 100%, for a median follow-up of 14.6 months
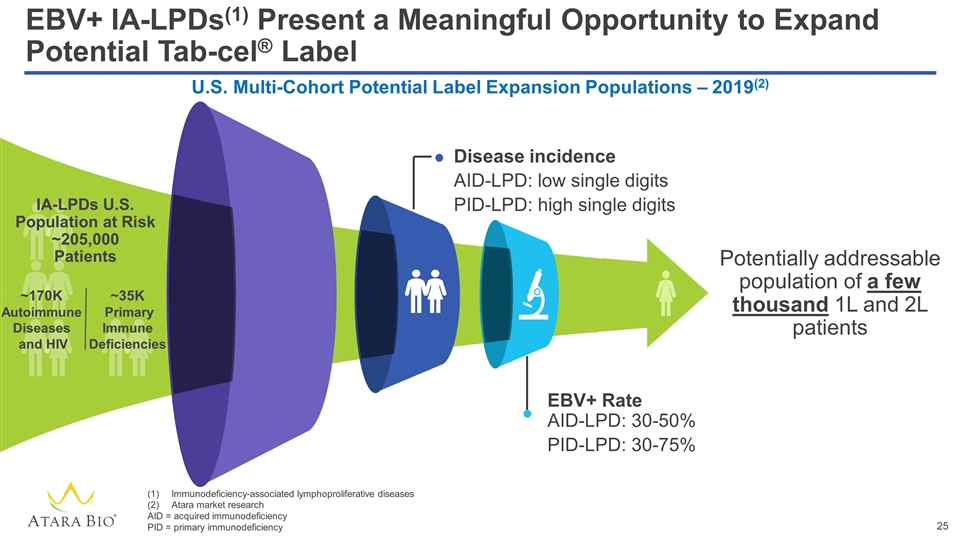
EBV+ IA-LPDs(1) Present a Meaningful Opportunity to Expand Potential Tab-cel® Label Potentially addressable population of a few thousand 1L and 2L patients Disease incidence AID-LPD: low single digits PID-LPD: high single digits EBV+ Rate AID-LPD: 30-50% PID-LPD: 30-75% U.S. Multi-Cohort Potential Label Expansion Populations – 2019(2) IA-LPDs U.S. Population at Risk ~205,000 Patients ~170K Autoimmune Diseases and HIV ~35K Primary Immune Deficiencies Immunodeficiency-associated lymphoproliferative diseases Atara market research AID = acquired immunodeficiency PID = primary immunodeficiency

Atara Strategic Priorities to Create Value: ATA188 ATA188 CAR T EBV T-cell immunotherapy for progressive multiple sclerosis (MS) Tab-cel® (tabelecleucel) Investigational T-cell immunotherapy for EBV-associated ultra-rare diseases FDA breakthrough designation & EMA PRIME for EBV+ PTLD

Multiple Sclerosis (MS) is a Debilitating Disease of the Central Nervous System with Few Treatment Options High Unmet Need Remains for Patients with Progressive MS Ayden PTLD champion Large patient population ~2.3 million patients diagnosed and living with MS worldwide ~1 million MS patients worldwide have a progressive form of the disease (PMS) For patients with progressive MS, prognosis is poor with current treatment options Current therapies modestly delay progression but do not fundamentally alter its course Growing evidence that EBV has a major role in the pathogenesis of MS Prior EBV infection is necessary for a patient to develop MS (1)(2) MS may be mediated by B cells that are infected with EBV (3) Ascherio A et al, Nat Rev Neurol. 2012;8:602-612. Endriz, J. et al., Neurol. Neuroimmunol. Neuroinflamm. (2017) 4, e308 Pender et al, Clin Transl Immunology. 2017;6(1):e126. Cencioni et al, Immunology. 2017;152:660–676 Harley et al, Nature Genetics 2018 Dan MS champion
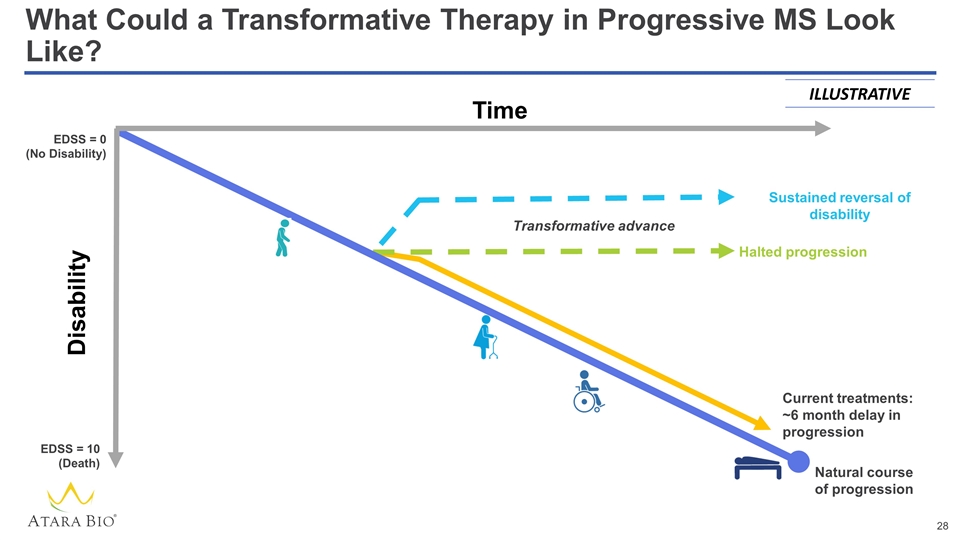
What Could a Transformative Therapy in Progressive MS Look Like? Sustained reversal of disability ILLUSTRATIVE Time EDSS = 0 (No Disability) EDSS = 10 (Death) Current treatments: ~6 month delay in progression Natural course of progression Transformative advance Halted progression Disability

ATA188 is a Potentially Transformative MS Treatment Halt or Reverses disability progression Safe and Well-tolerated Penetrates CNS Targets specific B cells and plasma cells Addresses underlying biology Profile of a Transformative Treatment Sustained Disability Improvement (SDI) observed in Phase 1a Acceptable safety profile observed in Phase 1a EBV-specific T-cells can traffic and cross the blood brain barrier Precision targeting to limits off-target activity MS pathogenesis increasingly linked to EBV Safety Efficacy Activity Targeting Intervention ATA188 Characteristics ü ü ü ü ü We are Moving Toward Biologics-Like COGM and Supply Chain at Commercial Scale
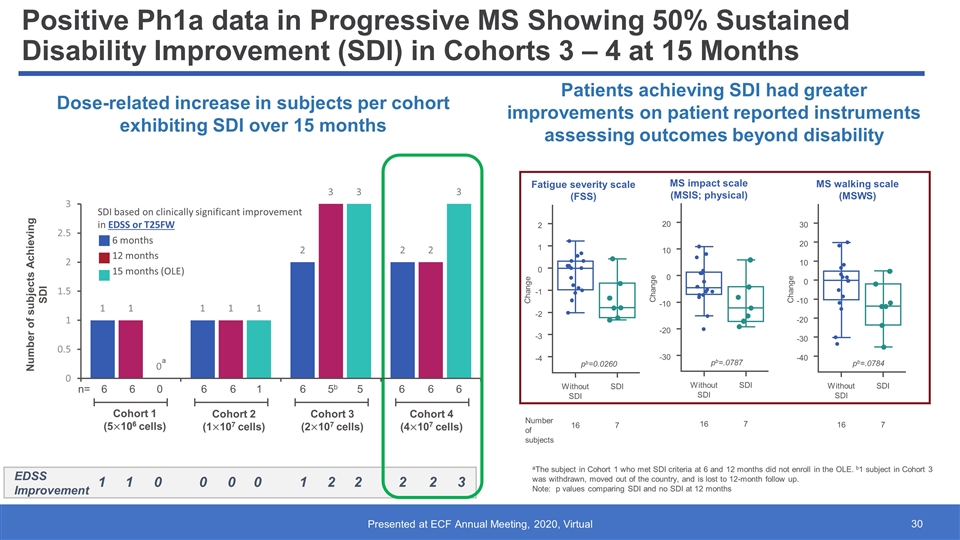
Positive Ph1a data in Progressive MS Showing 50% Sustained Disability Improvement (SDI) in Cohorts 3 – 4 at 15 Months Patients achieving SDI had greater improvements on patient reported instruments assessing outcomes beyond disability Dose-related increase in subjects per cohort exhibiting SDI over 15 months aThe subject in Cohort 1 who met SDI criteria at 6 and 12 months did not enroll in the OLE. b1 subject in Cohort 3 was withdrawn, moved out of the country, and is lost to 12-month follow up. Note: p values comparing SDI and no SDI at 12 months EDSS Improvement 1 1 1 2 2 2 0 0 Number of subjects Achieving SDI Cohort 1 (5106 cells) Cohort 2 (1107 cells) Cohort 3 (2107 cells) Cohort 4 (4107 cells) n= 6 6 6 6 6 5b 6 6 SDI based on clinically significant improvement in EDSS or T25FW 6 months 12 months 15 months (OLE) 0 1 5 6 a 0 0 2 3 2 1 0 -1 -2 -3 -4 Change 16 Without SDI 7 SDI Number of subjects pb=0.0260 Fatigue severity scale (FSS) MS impact scale (MSIS; physical) Change 20 10 0 -10 -20 -30 16 Without SDI 7 SDI pb=.0787 Change 30 20 10 -10 -20 -30 -40 16 Without SDI 7 SDI 0 pb=.0784 MS walking scale (MSWS)
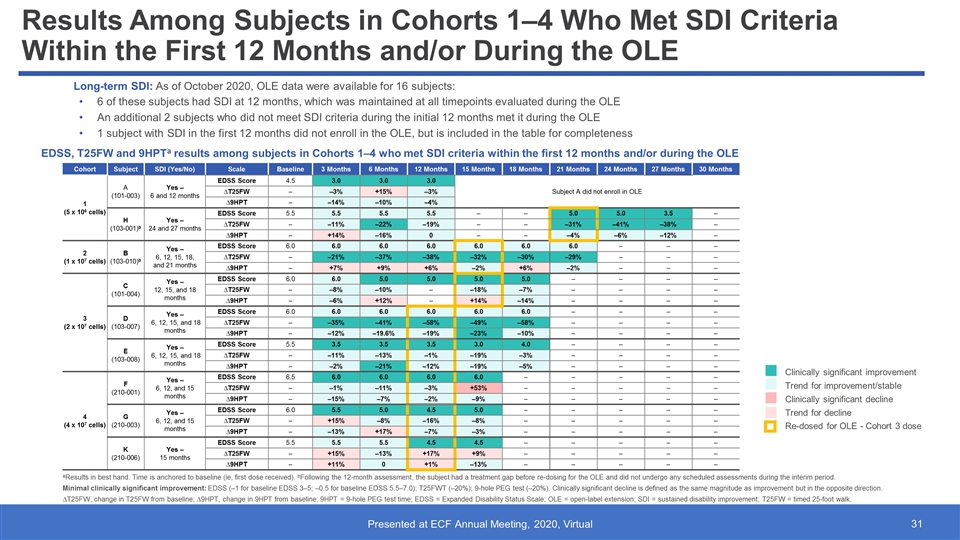
Results Among Subjects in Cohorts 1–4 Who Met SDI Criteria Within the First 12 Months and/or During the OLE EDSS, T25FW and 9HPTa results among subjects in Cohorts 1–4 who met SDI criteria within the first 12 months and/or during the OLE 6 of these subjects had SDI at 12 months, which was maintained at all timepoints evaluated during the OLE An additional 2 subjects who did not meet SDI criteria during the initial 12 months met it during the OLE 1 subject with SDI in the first 12 months did not enroll in the OLE, but is included in the table for completeness aResults in best hand. Time is anchored to baseline (ie, first dose received). bFollowing the 12-month assessment, the subject had a treatment gap before re-dosing for the OLE and did not undergo any scheduled assessments during the interim period. Minimal clinically significant improvement: EDSS (‒1 for baseline EDSS 3‒5; ‒0.5 for baseline EDSS 5.5‒7.0); T25FWT (‒20%); 9-hole PEG test (‒20%). Clinically significant decline is defined as the same magnitude as improvement but in the opposite direction. ∆T25FW, change in T25FW from baseline; ∆9HPT, change in 9HPT from baseline; 9HPT = 9-hole PEG test time; EDSS = Expanded Disability Status Scale; OLE = open-label extension; SDI = sustained disability improvement; T25FW = timed 25-foot walk. Cohort Subject SDI (Yes/No) Scale Baseline 3 Months 6 Months 12 Months 15 Months 18 Months 21 Months 24 Months 27 Months 30 Months 1 (5 x 106 cells) A (101-003) Yes – 6 and 12 months EDSS Score 4.5 3.0 3.0 3.0 Subject A did not enroll in OLE ∆T25FW ‒ ‒3% +15% ‒3% ∆9HPT ‒ ‒14% ‒10% ‒4% H (103-001)b Yes – 24 and 27 months EDSS Score 5.5 5.5 5.5 5.5 ‒ ‒ 5.0 5.0 3.5 ‒ ∆T25FW ‒ ‒11% ‒22% ‒19% ‒ ‒ ‒31% ‒41% ‒38% ‒ ∆9HPT ‒ +14% ‒16% 0 ‒ ‒ ‒4% ‒6% ‒12% ‒ 2 (1 x 107 cells) B (103-010)b Yes ‒ 6, 12, 15, 18, and 21 months EDSS Score 6.0 6.0 6.0 6.0 6.0 6.0 6.0 ‒ ‒ ‒ ∆T25FW ‒ –21% –37% –38% –32% –30% –29% ‒ ‒ ‒ ∆9HPT ‒ +7% +9% +6% –2% +6% –2% ‒ ‒ ‒ 3 (2 x 107 cells) C (101-004) Yes – 12, 15, and 18 months EDSS Score 6.0 6.0 5.0 5.0 5.0 5.0 ‒ ‒ ‒ ‒ ∆T25FW ‒ ‒8% ‒10% ‒ ‒18% ‒7% ‒ ‒ ‒ ‒ ∆9HPT ‒ ‒6% +12% ‒ +14% ‒14% ‒ ‒ ‒ ‒ D (103-007) Yes ‒ 6, 12, 15, and 18 months EDSS Score 6.0 6.0 6.0 6.0 6.0 6.0 ‒ ‒ ‒ ‒ ∆T25FW ‒ –35% –41% –58% –49% –58% ‒ ‒ ‒ ‒ ∆9HPT ‒ –12% –19.6% –19% –23% –10% ‒ ‒ ‒ ‒ E (103-008) Yes ‒ 6, 12, 15, and 18 months EDSS Score 5.5 3.5 3.5 3.5 3.0 4.0 ‒ ‒ ‒ ‒ ∆T25FW ‒ –11% –13% –1% –19% –3% ‒ ‒ ‒ ‒ ∆9HPT ‒ –2% –21% –12% –19% –5% ‒ ‒ ‒ ‒ 4 (4 x 107 cells) F (210-001) Yes – 6, 12, and 15 months EDSS Score 6.5 6.0 6.0 6.0 6.0 ‒ ‒ ‒ ‒ ‒ ∆T25FW ‒ ‒1% ‒11% ‒3% +53% ‒ ‒ ‒ ‒ ‒ ∆9HPT ‒ ‒15% ‒7% ‒2% ‒9% ‒ ‒ ‒ ‒ ‒ G (210-003) Yes – 6, 12, and 15 months EDSS Score 6.0 5.5 5.0 4.5 5.0 ‒ ‒ ‒ ‒ ‒ ∆T25FW ‒ +15% ‒8% ‒16% ‒8% ‒ ‒ ‒ ‒ ‒ ∆9HPT ‒ ‒13% +17% ‒7% ‒3% ‒ ‒ ‒ ‒ ‒ K (210-006) Yes – 15 months EDSS Score 5.5 5.5 5.5 4.5 4.5 ‒ ‒ ‒ ‒ ‒ ∆T25FW ‒ +15% ‒13% +17% +9% ‒ ‒ ‒ ‒ ‒ ∆9HPT ‒ +11% 0 +1% ‒13% ‒ ‒ ‒ ‒ ‒ Clinically significant improvement Trend for improvement/stable Clinically significant decline Trend for decline Re-dosed for OLE - Cohort 3 dose Long-term SDI: As of October 2020, OLE data were available for 16 subjects:
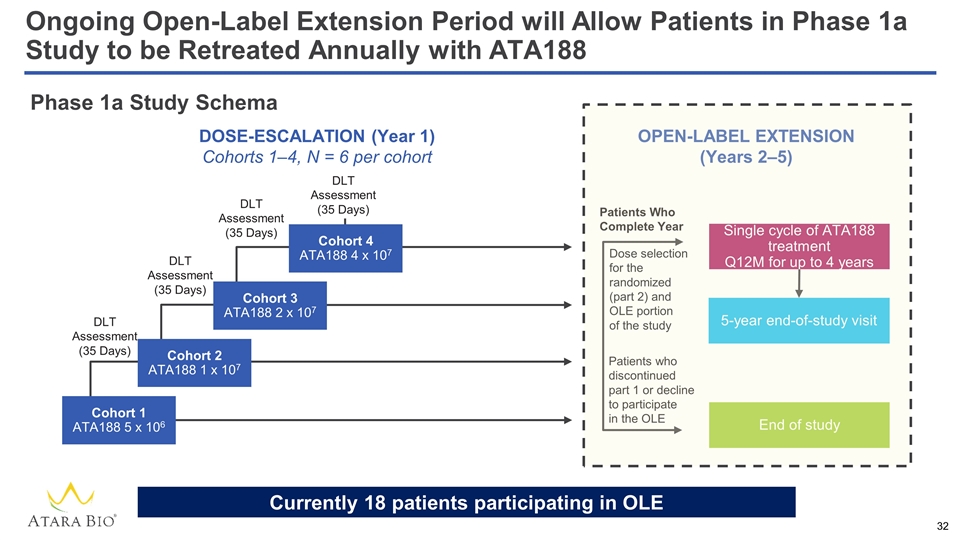
OPEN-LABEL EXTENSION (Years 2‒5) Single cycle of ATA188 treatment Q12M for up to 4 years 5-year end-of-study visit End of study Dose selection for the randomized (part 2) and OLE portion of the study Patients Who Complete Year Patients who discontinued part 1 or decline to participate in the OLE Phase 1a Study Schema Cohort 1 ATA188 5 x 106 Cohort 2 ATA188 1 x 107 Cohort 3 ATA188 2 x 107 Cohort 4 ATA188 4 x 107 DLT Assessment (35 Days) DLT Assessment (35 Days) DLT Assessment (35 Days) DLT Assessment (35 Days) DOSE-ESCALATION (Year 1) Cohorts 1–4, N = 6 per cohort Ongoing Open-Label Extension Period will Allow Patients in Phase 1a Study to be Retreated Annually with ATA188 Currently 18 patients participating in OLE

We Have Made Significant Progress with the FDA and are Advancing our Phase 2 RCT for ATA188, with an Interim Analysis Planned in H1 2022 FDA Registrational Feedback Received for Current RCT Primary endpoint: measuring disability improvement is acceptable; FDA preference for EDSS Study duration should be at least 12 months and T25FW can be used as a supportive measure Patient Population: our definition of non-active SPMS and non-active PPMS is acceptable Next Steps: We plan to have additional dialogue with FDA on how to approach the target population in order to potentially amend the RCT for registrational purposes, and to apply for expedited pathways for development Key Modifications Implemented in Amendment to Ongoing Phase 2 RCT Include EDSS as the primary endpoint and increase the sample size to 80 Planned Interim Analysis on Track We plan to conduct a formal interim analysis in H1 2022, including efficacy and safety, to confirm current development strategy RCT = Radomized Controlled TrialSPMS = Secondary Progressive Multiple Sclerosis EDSS = Expanded Disability Status ScalePPMS = Primary Progressive Multiple Sclerosis T25FW = Timed 25-foot walk

Multi-Billion Dollar Potential for a Transformative Therapy in Progressive MS Transformative Therapy: ~25% - 50% Higher Treatment Rate1 ~$5B - $7B PMS Overall Market Size Potential Annual US Revenue Opportunity in PMS of ~$3.5B+ Estimated US Market Forecast for Progressive MS (2025) ~45%+ Share for Best-in-Class Treatment2 43% Anti-CD20s 5 YR CAGR: up to ~9% Other therapies 57% ’20 ’25 Sources: 2019 GlobalData MS Market Forecast; 2019 Decision Research Group MS Landscape and Market Forecast; Atara Analysis 1 Based on reaching RRMS treatment level 2 Based on current anti-CD20 therapies or better Each 10% of share = ~$750M - $1B in Revenue

Atara Strategic Priorities to Create Value: CAR T CAR T Tab-cel® (tabelecleucel) Investigational T-cell immunotherapy for EBV-associated ultra-rare diseases FDA breakthrough designation & EMA PRIME for EBV+ PTLD ATA188 EBV T-cell immunotherapy for progressive multiple sclerosis (MS) ATA2271/ATA3271 (Solid Tumors) ATA3219 (B-cell malignancies) Other CAR T
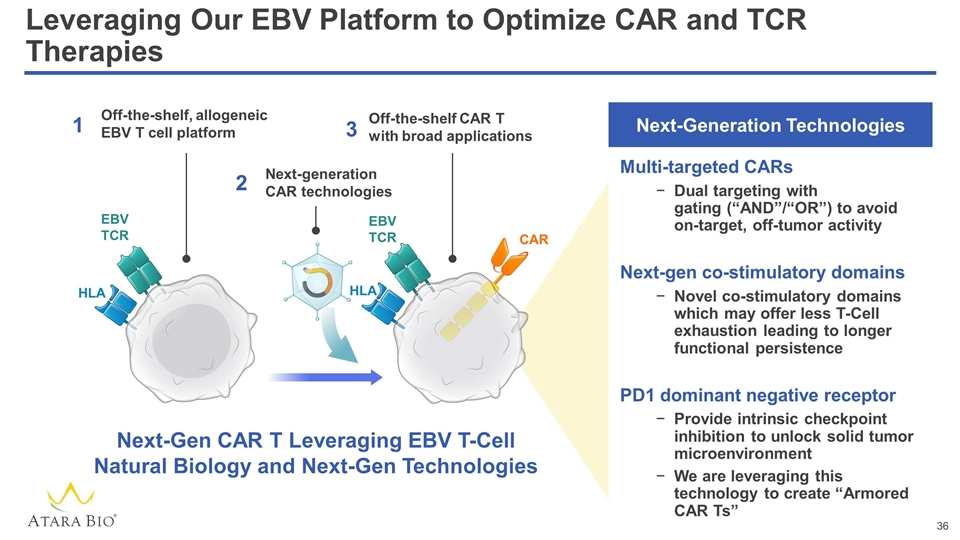
Leveraging Our EBV Platform to Optimize CAR and TCR Therapies Off-the-shelf, allogeneic EBV T cell platform Next-generation CAR technologies Off-the-shelf CAR T with broad applications 1 2 3 EBV TCR HLA EBV TCR HLA Next-Gen CAR T Leveraging EBV T-Cell Natural Biology and Next-Gen Technologies CAR Multi-targeted CARs Dual targeting with gating (“AND”/“OR”) to avoid on-target, off-tumor activity Next-gen co-stimulatory domains Novel co-stimulatory domains which may offer less T-Cell exhaustion leading to longer functional persistence PD1 dominant negative receptor Provide intrinsic checkpoint inhibition to unlock solid tumor microenvironment We are leveraging this technology to create “Armored CAR Ts” Next-Generation Technologies

Recognizes the leading position of Atara’s technology platform and capabilities in allogeneic cell therapy Agreement is fundamental element of Bayer’s new Cell & Gene Therapy strategy Bayer brings significant development & commercialization capabilities in oncology solid tumors, which complements Atara’s leading allogeneic T-cell platform We believe this collaboration maximizes the opportunity for ATA3271, a novel CAR T with PD-1 DNR and 1XX costimulatory domain which has the potential to be a first-in-class treatment with an optimized design for solid tumors Atara Biotherapeutics and Bayer Enter Strategic Collaboration for Mesothelin-Targeted CAR T Cell Therapies For Solid Tumors Atara will lead IND-enabling studies and process development for ATA3271 while Bayer will be responsible for submitting the IND and subsequent clinical development and commercialization As part of the transaction, Atara will also provide translational and clinical manufacturing services to be reimbursed by Bayer Atara will receive $60M in cash upon signing and is eligible to receive up to $610M in development, regulatory, and commercial milestone payments, plus tiered royalties up to low double-digit percentage of net sales Worldwide license agreement and research, development and manufacturing collaboration to develop Atara’s allogeneic off-the-shelf mesothelin CAR T program (ATA3271) and autologous program (ATA2271)
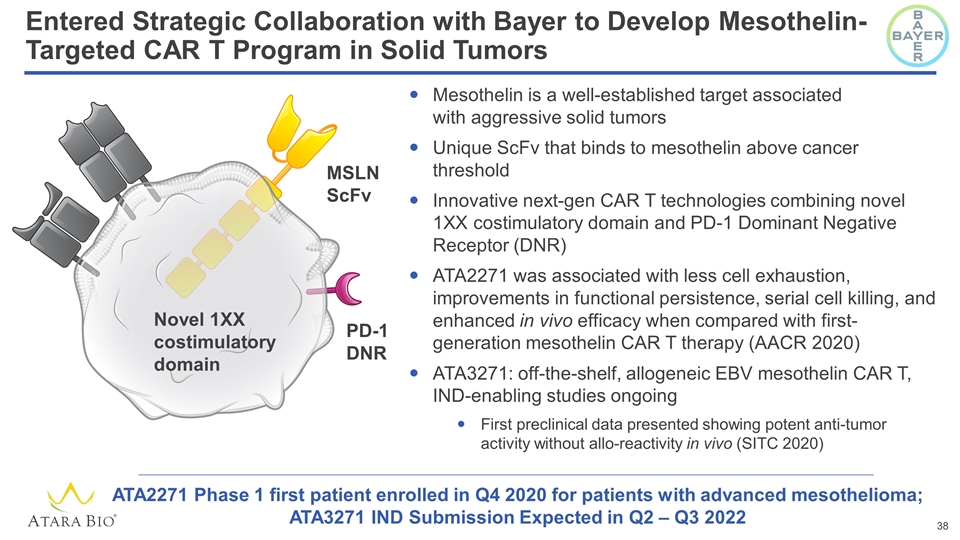
Entered Strategic Collaboration with Bayer to Develop Mesothelin-Targeted CAR T Program in Solid Tumors Mesothelin is a well-established target associated with aggressive solid tumors Unique ScFv that binds to mesothelin above cancer threshold Innovative next-gen CAR T technologies combining novel 1XX costimulatory domain and PD-1 Dominant Negative Receptor (DNR) ATA2271 was associated with less cell exhaustion, improvements in functional persistence, serial cell killing, and enhanced in vivo efficacy when compared with first-generation mesothelin CAR T therapy (AACR 2020) ATA3271: off-the-shelf, allogeneic EBV mesothelin CAR T, IND-enabling studies ongoing First preclinical data presented showing potent anti-tumor activity without allo-reactivity in vivo (SITC 2020) MSLN ScFv Novel 1XX costimulatory domain PD-1 DNR ATA2271 Phase 1 first patient enrolled in Q4 2020 for patients with advanced mesothelioma; ATA3271 IND Submission Expected in Q2 – Q3 2022
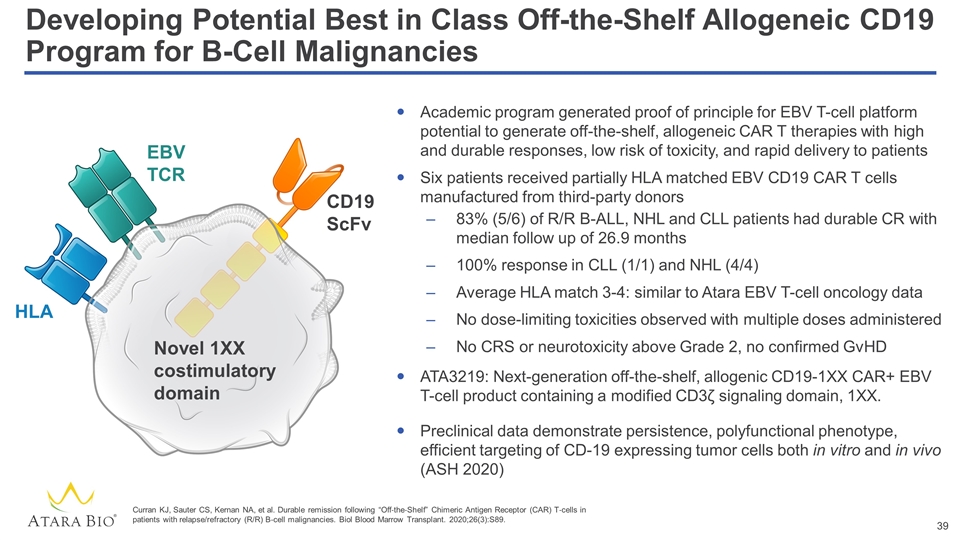
Developing Potential Best in Class Off-the-Shelf Allogeneic CD19 Program for B-Cell Malignancies Academic program generated proof of principle for EBV T-cell platform potential to generate off-the-shelf, allogeneic CAR T therapies with high and durable responses, low risk of toxicity, and rapid delivery to patients Six patients received partially HLA matched EBV CD19 CAR T cells manufactured from third-party donors 83% (5/6) of R/R B-ALL, NHL and CLL patients had durable CR with median follow up of 26.9 months 100% response in CLL (1/1) and NHL (4/4) Average HLA match 3-4: similar to Atara EBV T-cell oncology data No dose-limiting toxicities observed with multiple doses administered No CRS or neurotoxicity above Grade 2, no confirmed GvHD ATA3219: Next-generation off-the-shelf, allogenic CD19-1XX CAR+ EBV T-cell product containing a modified CD3ζ signaling domain, 1XX. Preclinical data demonstrate persistence, polyfunctional phenotype, efficient targeting of CD-19 expressing tumor cells both in vitro and in vivo (ASH 2020) CD19 ScFv Novel 1XX costimulatory domain EBV TCR HLA Curran KJ, Sauter CS, Kernan NA, et al. Durable remission following “Off‐the‐Shelf” Chimeric Antigen Receptor (CAR) T‐cells in patients with relapse/refractory (R/R) B‐cell malignancies. Biol Blood Marrow Transplant. 2020;26(3):S89.

Thank You

Appendix
Tab-cel®
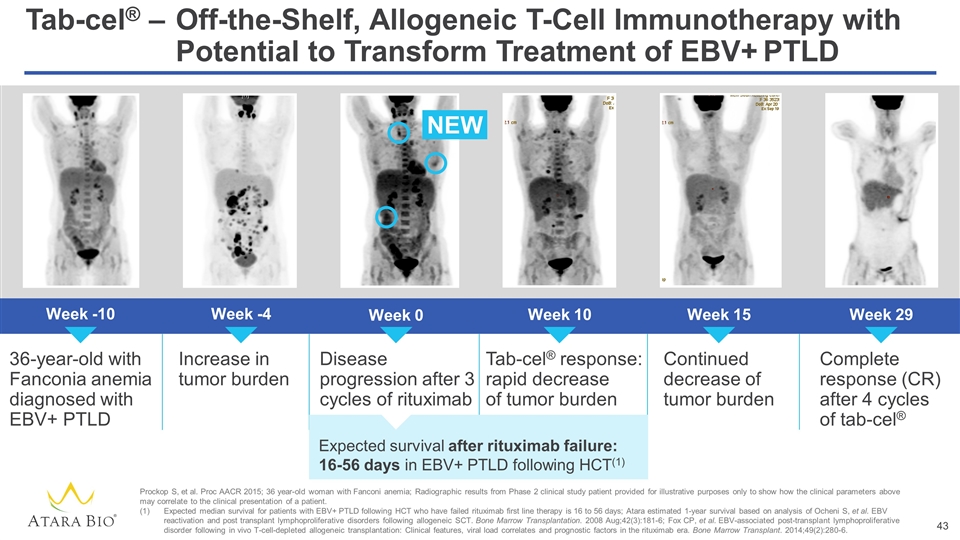
Tab-cel® –Off-the-Shelf, Allogeneic T-Cell Immunotherapy with Potential to Transform Treatment of EBV+ PTLD Week -10 36-year-old with Fanconia anemia diagnosed with EBV+ PTLD Week -4 Week 10 Week 15 Week 0 Increase in tumor burden Disease progression after 3 cycles of rituximab Tab-cel® response: rapid decrease of tumor burden Continued decrease of tumor burden Complete response (CR) after 4 cycles of tab-cel® Week 29 NEW Expected survival after rituximab failure: 16-56 days in EBV+ PTLD following HCT(1) Prockop S, et al. Proc AACR 2015; 36 year-old woman with Fanconi anemia; Radiographic results from Phase 2 clinical study patient provided for illustrative purposes only to show how the clinical parameters above may correlate to the clinical presentation of a patient. Expected median survival for patients with EBV+ PTLD following HCT who have failed rituximab first line therapy is 16 to 56 days; Atara estimated 1-year survival based on analysis of Ocheni S, et al. EBV reactivation and post transplant lymphoproliferative disorders following allogeneic SCT. Bone Marrow Transplantation. 2008 Aug;42(3):181-6; Fox CP, et al. EBV-associated post-transplant lymphoproliferative disorder following in vivo T-cell-depleted allogeneic transplantation: Clinical features, viral load correlates and prognostic factors in the rituximab era. Bone Marrow Transplant. 2014;49(2):280-6.
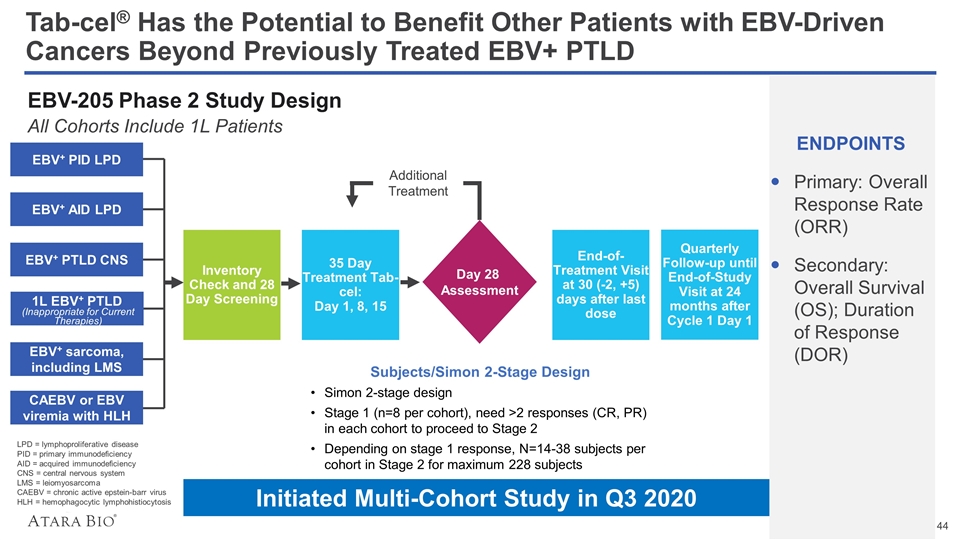
Tab-cel® Has the Potential to Benefit Other Patients with EBV-Driven Cancers Beyond Previously Treated EBV+ PTLD EBV-205 Phase 2 Study Design All Cohorts Include 1L Patients ENDPOINTS Primary: Overall Response Rate (ORR) Secondary: Overall Survival (OS); Duration of Response (DOR) Inventory Check and 28 Day Screening 35 Day Treatment Tab-cel: Day 1, 8, 15 Subjects/Simon 2-Stage Design Simon 2-stage design Stage 1 (n=8 per cohort), need >2 responses (CR, PR) in each cohort to proceed to Stage 2 Depending on stage 1 response, N=14-38 subjects per cohort in Stage 2 for maximum 228 subjects EBV+ PID LPD EBV+ AID LPD EBV+ PTLD CNS 1L EBV+ PTLD EBV+ sarcoma, including LMS CAEBV or EBV viremia with HLH Day 28 Assessment End-of-Treatment Visit at 30 (-2, +5) days after last dose Quarterly Follow-up until End-of-Study Visit at 24 months after Cycle 1 Day 1 Additional Treatment (Inappropriate for Current Therapies) LPD = lymphoproliferative disease PID = primary immunodeficiency AID = acquired immunodeficiency CNS = central nervous system LMS = leiomyosarcoma CAEBV = chronic active epstein-barr virus HLH = hemophagocytic lymphohistiocytosis Initiated Multi-Cohort Study in Q3 2020
ATA 188
ATA188 Off-the-shelf T cells delivered from inventory Phase 1 trial successfully demonstrated safety and No pretreatment required in the clinical trial protocol Precision targeting to select EBV antigens limits off-target activity Two-hour monitoring following 5-10 minute IV infusion Administered as an outpatient therapy A bold vision to transform MS therapy Potential for improvement of disease in progressive MS
The Role of EBV in Multiple Sclerosis
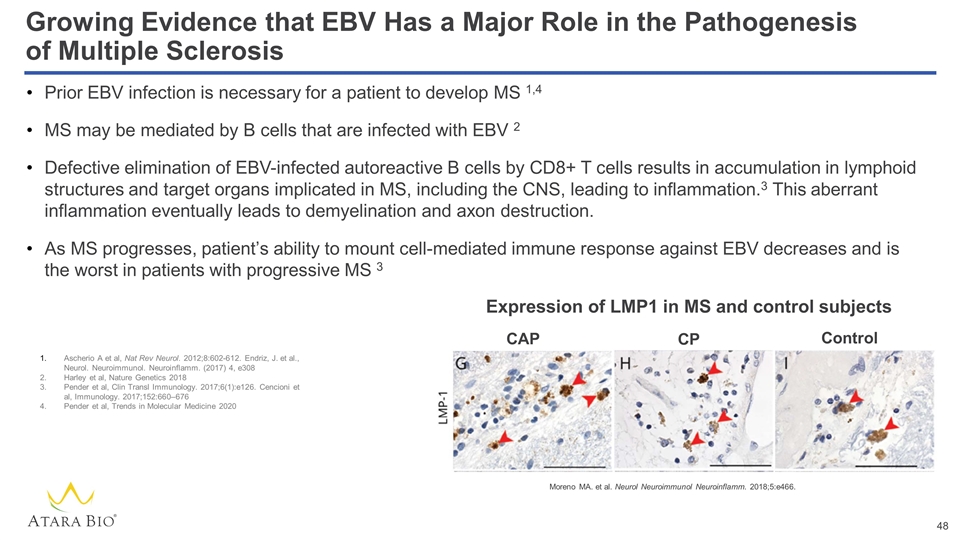
Prior EBV infection is necessary for a patient to develop MS 1,4 MS may be mediated by B cells that are infected with EBV 2 Defective elimination of EBV-infected autoreactive B cells by CD8+ T cells results in accumulation in lymphoid structures and target organs implicated in MS, including the CNS, leading to inflammation.3 This aberrant inflammation eventually leads to demyelination and axon destruction. As MS progresses, patient’s ability to mount cell-mediated immune response against EBV decreases and is the worst in patients with progressive MS 3 Growing Evidence that EBV Has a Major Role in the Pathogenesis of Multiple Sclerosis Moreno MA. et al. Neurol Neuroimmunol Neuroinflamm. 2018;5:e466. Ascherio A et al, Nat Rev Neurol. 2012;8:602-612. Endriz, J. et al., Neurol. Neuroimmunol. Neuroinflamm. (2017) 4, e308 Harley et al, Nature Genetics 2018 Pender et al, Clin Transl Immunology. 2017;6(1):e126. Cencioni et al, Immunology. 2017;152:660–676 Pender et al, Trends in Molecular Medicine 2020 Expression of LMP1 in MS and control subjects CAP CP Control
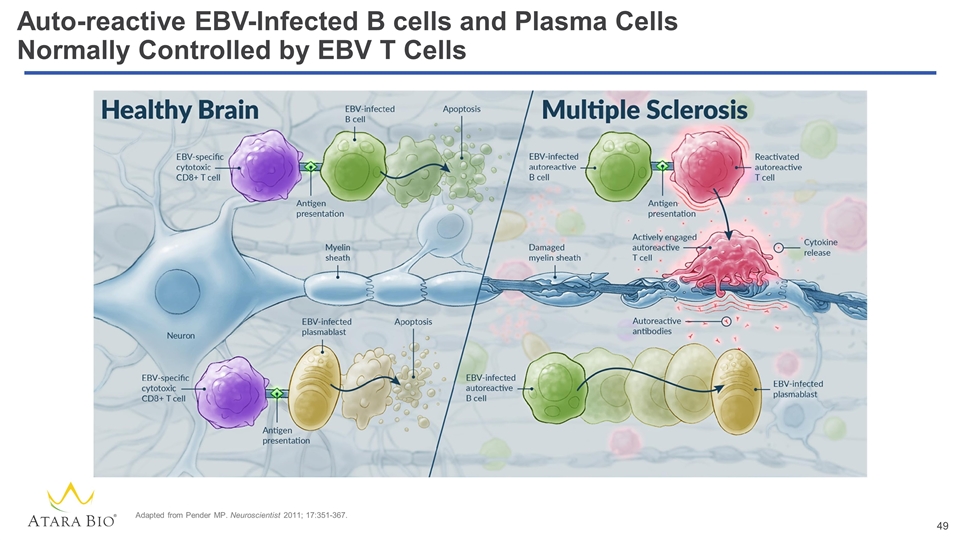
Auto-reactive EBV-Infected B cells and Plasma Cells Normally Controlled by EBV T Cells Adapted from Pender MP. Neuroscientist 2011; 17:351-367.
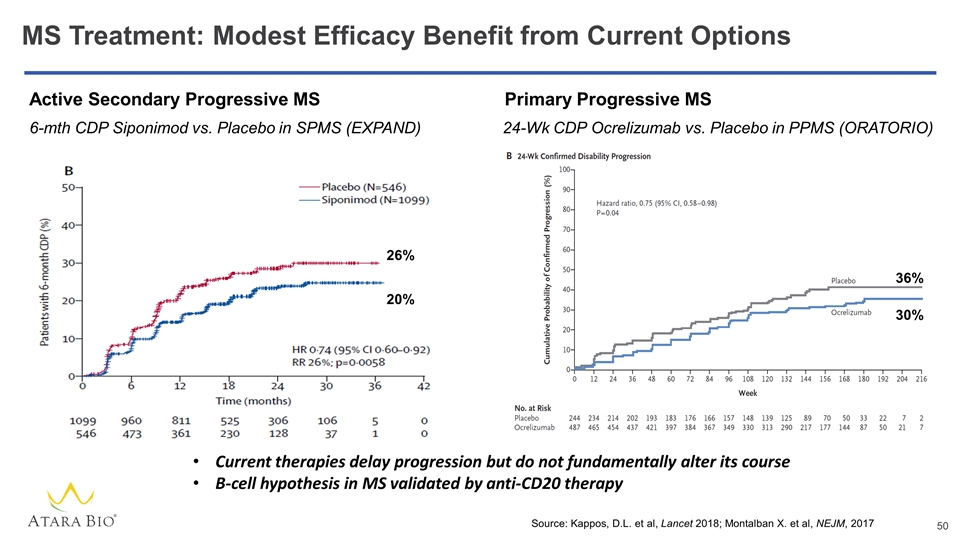
MS Treatment: Modest Efficacy Benefit from Current Options 26% 20% 6-mth CDP Siponimod vs. Placebo in SPMS (EXPAND) Source: Kappos, D.L. et al, Lancet 2018; Montalban X. et al, NEJM, 2017 Current therapies delay progression but do not fundamentally alter its course B-cell hypothesis in MS validated by anti-CD20 therapy Active Secondary Progressive MS 36% 30% 24-Wk CDP Ocrelizumab vs. Placebo in PPMS (ORATORIO) Primary Progressive MS

Based on Encouraging Clinical Data, We Have Increased our Investment in the ATA188 Program ATA188 Investment Summary Planned expansion to at least 80 patients in Phase 2 double-blind placebo-controlled study Plan to change primary endpoint to EDSS improvement endpoint while maintaining other disability improvement and biological endpoints as secondary Additional biomarker studies (including MOA) Novel stirred-tank bioreactor manufacturing scale-up
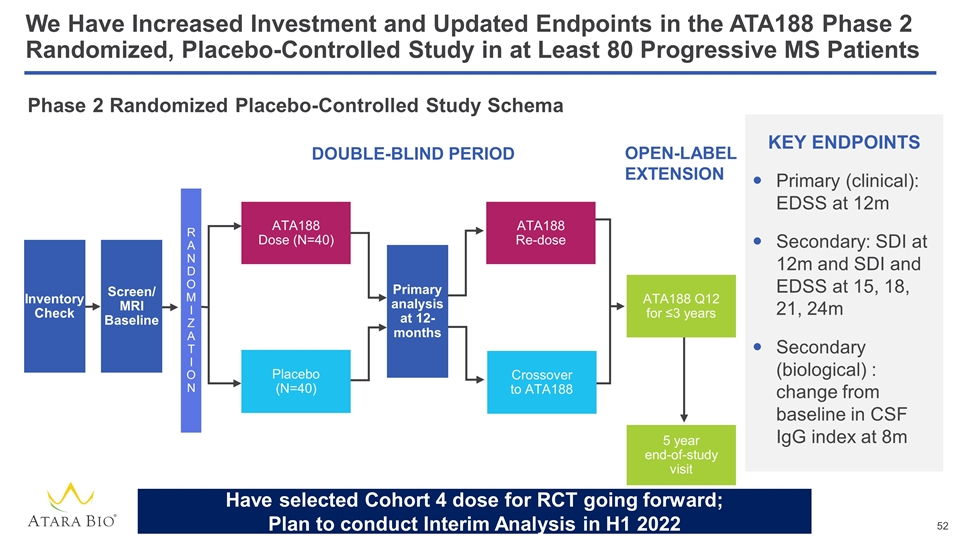
We Have Increased Investment and Updated Endpoints in the ATA188 Phase 2 Randomized, Placebo-Controlled Study in at Least 80 Progressive MS Patients Phase 2 Randomized Placebo-Controlled Study Schema DOUBLE-BLIND PERIOD OPEN-LABEL EXTENSION KEY ENDPOINTS Primary (clinical): EDSS at 12m Secondary: SDI at 12m and SDI and EDSS at 15, 18, 21, 24m Secondary (biological) : change from baseline in CSF IgG index at 8m Inventory Check ATA188 Dose (N=40) Screen/ MRI Baseline R A N D O M I Z A T I O N Placebo (N=40) ATA188 Re-dose Crossover to ATA188 ATA188 Q12 for ≤3 years 5 year end-of-study visit Primary analysis at 12-months Have selected Cohort 4 dose for RCT going forward; Plan to conduct Interim Analysis in H1 2022
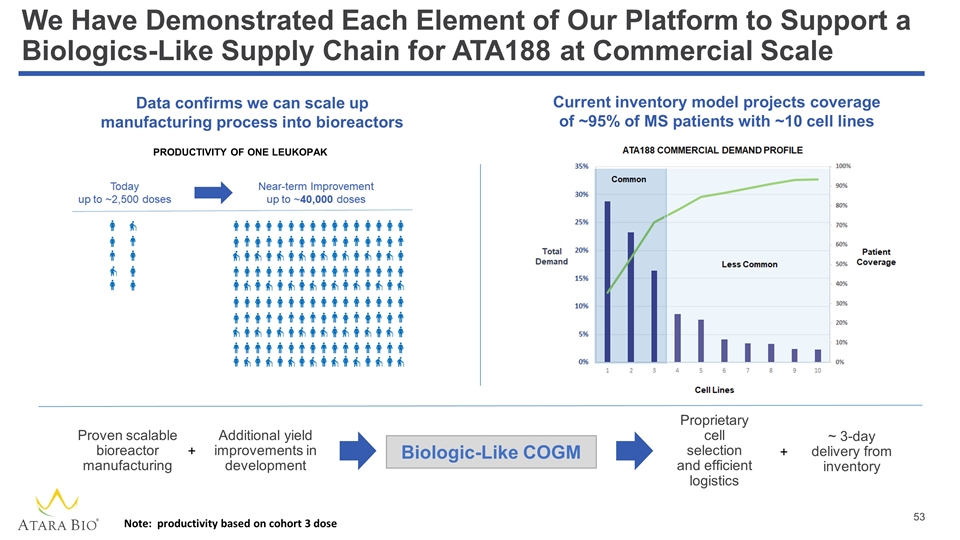
We Have Demonstrated Each Element of Our Platform to Support a Biologics-Like Supply Chain for ATA188 at Commercial Scale PRODUCTIVITY OF ONE LEUKOPAK Proven scalable bioreactor manufacturing Additional yield improvements in development Proprietary cell selection and efficient logistics ~ 3-day delivery from inventory + + Current inventory model projects coverage of ~95% of MS patients with ~10 cell lines Data confirms we can scale up manufacturing process into bioreactors Biologic-Like COGM Note: productivity based on cohort 3 dose
CAR T Portfolio
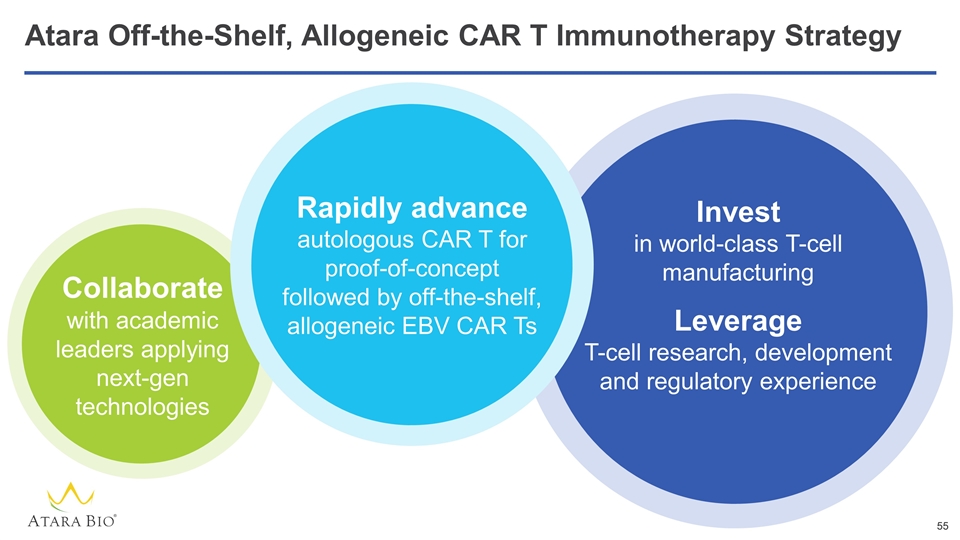
Atara Off-the-Shelf, Allogeneic CAR T Immunotherapy Strategy Invest in world-class T-cell manufacturing Leverage T-cell research, development and regulatory experience Collaborate with academic leaders applying next-gen technologies Rapidly advance autologous CAR T for proof-of-concept followed by off-the-shelf, allogeneic EBV CAR Ts

Exclusive License to Mesothelin-Targeted CAR T Immunotherapy for Solid Tumors from MSK Mesothelin is an attractive target associated with aggressive solid tumors Aberrant mesothelin expression promotes cancer cell proliferation and confers resistance to apoptosis Associated with mesothelioma, triple-negative breast cancer and non-small cell lung cancer Mesothelin-associated cancers(1) Incidence: ~340,000 patients Prevalence: ~2 million patients Morello A, Sadelain M, Adusumilli PS. Mesothelin-Targeted CARs: Driving T Cells to Solid Tumors. Cancer Discov. 2016 Feb;6(2):133-46; U.S. incidence/prevalence. Frequency and distribution pattern of the mesothelin protein in solid malignancies. (1,2)

Atara CAR T Pipeline – Applying Next-Generation Technologies in Collaboration with Academic Leaders AML: acute myeloid leukemia; DNR: Dominant Negative Receptor Worldwide license agreement and research, development and manufacturing collaboration with Bayer to develop Atara’s allogeneic off-the-shelf mesothelin CAR T program (ATA3271) and autologous program (ATA2271) Mesothelin is expressed at high levels on the surface of cells in aggressive solid tumors including mesothelioma, triple-negative breast cancer, esophageal cancer, pancreatic cancer and non-small cell lung cancer Indication Target CAR T Technologies ATA2271 Autologous Solid tumors(2) Mesothelin PD-1 DNR 1XX co-stimulation ATA3271 Off-the-shelf, allogeneic Solid tumors(2) Mesothelin PD-1 DNR 1XX co-stimulation ATA3219 Off-the-shelf, allogeneic B-cell malignancies CD19 1XX co-stimulation ATA2321 Autologous AML Dual-targeted undisclosed Mut06 co-stimulation ATA2431 Autologous B-cell malignancies CD19-CD20 Mut06 co-stimulation Other CAR T Infectious diseases Undisclosed 1XX co-stimulation (1) (1)